APPENDIX 1
CONVERSION FACTORS AND CONSTANTS
Conversion Factors (arranged alphabetically)
Acceleration (L t−2)1
- 1 m/sec2 = 3.2808 ft/sec2 = 39.3701 in./sec2
- 1 ft/sec2 = 0.3048 m/sec2 = 12.0 in./sec2
 = 9.80665 m/sec2 = 32.174 ft/sec2 (standard gravity constant at Earth's surface)
= 9.80665 m/sec2 = 32.174 ft/sec2 (standard gravity constant at Earth's surface)
Area (L2)
- 1 ft2 = 144.0 in.2 = 0.092903 m2
- 1 m2 = 1550.0 in.2 = 10.7639 ft2
- 1 in.2 = 6.4516 × 10−4 m2
Density (M L3)
- Specific gravity is dimensionless, but has the same numerical value as density expressed in g/cm3; 1 liter = 10−3 m3
- 1 kg/m3 = 6.24279 × 10−2 lbm/ft3 = 3.61273 × 10−5 lbm/in.3
- 1 lbm/ft3 = 16.0184 kg/m3
- 1 lbm/in.3 = 2.76799 × 104 kg/m3
Energy, also Work or Heat (M L2t−2)
- 1.0 Btu = 1055.056 J (joule)
- 1.0 kW‐hr = 3.600 × 106 J
- 1.0 ft‐lbf = 1.355817 J
- 1.0 cal = 4.1868 J
- 1.0 kcal = 4186.8 J
Force (M Lt‐ 2)
- 1.0 lbf = 4.448221 N
- 1 dyne = 10−5 N
- 1.0 kg (force) [used in Europe] = 9.80665 N
- 1.0 ton (force) [used in Europe] = 1000 kg (force)
- 1.0 N = 0.2248089 lbf
- 1.0 millinewton (mN) = 10−3 N
- Weight is the force acting on a mass being accelerated by gravity (
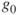 applies at the Earth's surface)
applies at the Earth's surface)
Length (L)
- 1 m = 3.2808 ft = 39.3701 in.
- 1 ft = 0.3048 m = 12.0 in.
- 1 in. = 2.540 cm = 0.0254 m
- 1 mile = 1.609344 km = 1609.344 m = 5280.0 ft
- 1 nautical mile = 1852.00 m
- 1 mil = 0.0000254 m = 1.00 × 10−3 in.
- 1 micron (µm) = 10−6 m
- 1 astronomical unit (au) = 1.49600 × 1011 m
Mass (M)
- 1 slug = 32.174 lbm
- 1 kg = 2.205 lbm = 1000 g
- 1 lbm = 16 ounces = 0.4536 kg
Power (M L2t−3)
- 1 Btu/sec = 0.2924 W (watt)
- 1 J/sec = 1.0 W = 0.001 kW
- 1 cal/sec = 4.186 W
- 1 horsepower = 550 ft‐lbf/sec = 745.6998 W
- 1 ft‐lbf/sec = 1.35581 W
Pressure (M L−1 t−2)
- 1 bar = 105 N/m2 = 0.10 MPa
- 1 atm = 0.101325 MPa = 14.696 psia
- 1 mm of mercury = 133.322 N/m2 (or Pa)
- 1 MPa = 106 N/m2
- 1 psi or lbf/in.2 = 6894.757 N/m2
Speed (or linear velocity) (L t−1)
- 1 ft/sec = 0.3048 m/sec = 12.00 in./sec
- 1 m/sec = 3.2808 ft/sec = 39.3701 in./sec
- 1 knot = 0.5144 m/sec
- 1 mile/hr = 0.4770 m/sec
Specific Heat (L2 t−2 T−1)
- 1 g‐cal/g‐°C = 1 kg‐cal/kg‐K = 1 Btu/lbm‐°F = 4.186 J/g‐°C = 1.163 × 10−3 kW‐hr/kg‐K
Temperature (T)
- 1 K = (9/5)°R = 1.80°R
- 0°C = 273.15 K
- 0°F = 459.67°R
- °C = (5/9)(°F − 32) and °F = (9/5)°C + 32
Time (t)
- 1 mean solar day = 24 hr = 1440 min = 86, 400 sec
- 1 calendar year = 365 days = 3.1536 × 107 sec
Viscosity (M L−1 t−1)
- 1 centistoke = 1.00 × 10−6 m2/sec (kinematic viscosity)
- 1 centipoise = 1.00 × 10−3 kg/m sec
- 1 lbf‐sec/ft2 = 47.88025 kg/m sec
Constants
| Mechanical equivalent of heat = 4.186 joule/cal = 777.9 ft‐lbf/Btu = 1055 joule/Btu | |||
| Universal gas constant = 8314.3 J/kg‐mol‐K = 1545 ft‐lbf/lbm‐mol‐°R | |||
| Molecular volume of an ideal gas = 22.41 liter/kg‐mol at standard conditions | |||
| Electron charge = 1.60218 × 10−19 coulomb | |||
| Permittivity of vacuum = 8.854187 × 10−12 farad/m | |||
| Gravitational constant = 6.674 × 10−11 m3/kg‐sec2 | |||
| Boltzmann's constant | 1.38065003 × 10−23 J/K | ||
| Electron mass | 9.109381 × 10−31 kg | ||
| Avogadro's number | 6.02252 × 1026/kg‐mol | ||
| Stefan–Boltzmann constant | 5.6696 × 10−8 W/m2‐K−4 | ||
Notes
..................Content has been hidden....................
You can't read the all page of ebook, please click here login for view all page.
